Chemical Reactions and Equations Class 10 MCQ is one of the best strategies to prepare for the CBSE Class 10 Board exam. If you want to complete a grasp concept or work on one’s score, there is no method except constant practice. Students can improve their speed and accuracy by doing more Chemical Reactions and Equations class 10, which will help them all through their board test.
Chemical Reactions and Equations Class 10 MCQ Questions with Answers
Class 10 Science MCQ with answers are given here to chapter 1 Chemical Reactions and Equations. These MCQs are based on the latest CBSE board syllabus and relate to the latest Class 10 Science syllabus. By Solving these Class 10 MCQs, you will be able to analyze all of the concepts quickly in the chapter and get ready for the Class 10 Annual exam.
Learn Chemical Reactions and Equations Class 10 MCQ with answers pdf free download according to the latest CBSE and NCERT syllabus. Students should prepare for the examination by solving CBSE Class 10 Chemical Reactions and Equations MCQ with answers given below.
Question 1: Which of the following is a decomposition reaction?
(a) 2HgO Heat→2Hg + O2
(b) CaCO3 Heat→CaO + CO2
(c) 2H2O Electrolysi →H2 + O2
(d) All of these
Answer
D
Question 2: In which of the following the identity of initial substance remains unchanged?
(a) Curdling of milk
(b) Formation of crystals by process of crystallisation
(c) Fermentation of grapes
(d) Digestion of food
Answer
B
Question 3: On the basis of following features, identify the correct option.
(i) This reaction occurs during corrosion.
(ii) This reaction occurs during respiration.
(a) Decomposition reaction
(b) Redox reaction
(c) Combination reaction
(d) Endothermic reaction
Answer
B
Question 4: Which of the following is not a physical change?
(a) Boiling of water to give water vapour.
(b) Melting of ice to give water.
(c) Dissolution of salt in water.
(d) Combustion of Liquefied Petroleum Gas (LPG).
Answer
D
Question 5: What happens when copper rod is dipped in iron sulphate solution?
(a) Copper displaces iron
(b) Blue colour of copper sulphate solution is obtained
(c) No reaction takes place
(d) Reaction is exothermic
Answer
C
Question 6: A dilute solution of sodium carbonate was added to two test tubes (A) containing dil HCl and (B) containing dilute NaOH. The correct observation was –
(a) a brown coloured gas liberated in test tube A.
(b) a brown coloured gas liberated in test tube B.
(c) a colourless gas liberated in test tube A.
(d) a colourless gas liberated in test tube B.
Answer
C
Question 7: When hydrogen sulphide gas is passed through a blue solution of copper sulphate, a black precipitate of copper sulphide is obtained and the sulphuric acid so formed remains in the solution. The reaction is an example of –
(a) a combination reaction
(b) a displacement reaction
(c) a decomposition reaction
(d) a double decomposition reaction
Answer
D
Question 8: Which of the following reactions involves the combination of two elements?
(a) CaO + CO2 → CaCO3
(b) 4Na + O2 → 2Na2O
(c) SO2 + 1/2 O2→ SO3
(d) NH3 + HCl → NH4Cl
Answer
B
Question 9: Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
Answer
C
Question 10: A student added dilute HCl to a test tube containing zinc granules and made following observations which one is correct?
(a) The zinc surface became dull and black.
(b) A gas evolved which burns with a pop sound.
(c) The solution remained colourless.
(d) The solution becomes green in colour.
Answer
B
Question 11 : The reaction in which two compounds exchange their ions to form two new compounds is –
(a) a displacement reaction
(b) a decomposition reaction
(c) an isomerization reaction
(d) a double displacement reaction
Answer
D
Question 12 : A balanced chemical equation is in accordance with –
(a) Avogadro’s law
(b) law of multiple proportion
(c) law of conservation of mass
(d) law of gaseous volumes.
Answer
C
Question 13 : The schematic diagram is given below

Which of the following is a correct statement ?
(a) A and E are chemically same.
(b) A and D are chemically same.
(c) D and E are chemically same.
(d) C and E are chemically same.
Answer
B
Question 14 : CuO + H2 → H2O + Cu, reaction is an example of –
(a) redox reaction
(b) synthesis reaction
(c) neutralisation
(d) analysis reaction
Answer
A
Question 15. Which of the following is a displacement reaction?
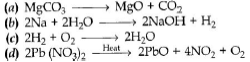
Answer
B
Question 16: When the gases sulphur dioxide and hydrogen sulphide mix in the presence of water, the reaction is SO2 + 2H2S → 2H2O + 2S. Here hydrogen sulphide is acting as –
(a) an oxidising agent
(b) a reducing agent
(c) a dehydrating agent
(d) a catalyst
Answer
B
Question 17: Zn+ H2SO4(dil) → ZnSO4 + H2↑ Above reaction is –
(a) decomposition reaction
(b) single displacement reaction
(c) combination reaction
(d) synthesis reaction
Answer
B
Question 18. Which of the following are exothermic processes?
(i) Reaction of water with quick lime
(ii) Dilution of an acid
(iii) Evaporation of water
(iv) Sublimation of camphor (crystals)
(a) (i) and (ii)
(b) (ii) and (iii)
(c) (i) and (iv)
(d) (ii) and (iv)
Answer
A
Question 19. Oxidation is a process which involves
(i) addition of oxygen
(ii) addition of hydrogen
(iii) removal of oxygen
(iv) removal of hydrogen
(a) (i) and (ii)
(b) (ii) and (iii)
(c) (i) and (iv)
(d) (ii) and (iv)
Answer
C
Question 20. Magnesium ribbon is rubbed before burning because it has a coating of
(a) basic magnesium carbonate
(b) basic magnesium oxide
(c) basic magnesium sulphide
(d) basic magnesium chloride
Answer
B
Question 21. Which of the following statements about the given reaction are correct?
3Fe (s) + 4H2O (g) → Fe3O4 (s) + 4 H2 (g)
(i) Iron metal is getting oxidized
(ii) Water is getting reduced
(iii) Water is acting as reducing agent
(iv) Water is acting as oxidizing agent
(a) (i), (ii) and (iii)
(b) (ii) and (iv)
(c) (i), (ii) and (iv)
(d) (ii) and (iv)
Answer
C
Question 22. An example of a chemical change is _____.
(1) formation of clouds.
(2) glowing of an electric light.
(3) dropping sodium into water.
(4) dissolving of salt in water.
Answer
3
Question 23. Physical changes are usually _______ in nature.
(1) temporary
(2) permanent
(3) irreversible
(4) endothermic
Answer
1
Question 24. Which of the following are chemical changes ?
(i) Digestion of food
(ii) Liquefaction of air
(iii) Ripening of fruit
(iv) Dissolution of sulphur in carbon disulphide
(v) Freezing of water
(vi) Electrolysis of water
(1) (i) to (iv) all
(2) (i), (iii) & (vi)
(3) (i), (iii) & (iv)
(4) (iii), (iv) & (vi)
Answer
2
Question 25. Which among the following is not a physical change?
(1) Melting of solid to liquid.
(2) Vaporisation of liquids to gas.
(3) Liquefaction of gases to liquid.
(4) Decay of matter.
Answer
4
Question 26. Which of the following is a combustion reaction ?
(1) Boiling of water
(2) Melting of wax
(3) Burning of petrol
(4) None of these
Answer
4
Question 27. The reaction, H2 + Cl2 →2HCl is
(1) an oxidation reaction
(2) a reduction reaction
(3) a combination reaction
(4) an isomerisation reaction
Answer
3
Question 28. Which among the following is not a chemical change?
(1) Melting of ice.
(2) Carbon cycle.
(3) Dehydration of substances.
(4) Fermentation of substances.
Answer
1
Question 29. Decomposition of water is
(1) electrolytic
(2) thermal
(3) photolytic
(4) All of the above
Answer
1
Question 30. Which of these will cause a chemical change to occur ?
(1) Grinding of wheat into flour.
(2) Lighting of a gas stove.
(3) Evaporation of water from a lake.
(4) Ringing of an electric bell.
Answer
2
Question 31. Chemical changes are __________.
(1) temporary, reversible and a new substance is produced.
(2) always accompanied by exchange of light.
(3) permanent, irreversible and a new substance is produced.
(4) never accompanied by exchange of light and heat energy.
Answer
3
Question 32. Which of the following is a decomposition reaction ?
(1) NaOH + HCl → NaCl + H2O
(2) NH4CNO → H2NCONH2
(3) 2KClO3 → 2KCl + 3O2
(4) H2 + I2 → 2Hl
Answer
3
Question 33. Which of the following is not a decomposition reaction ?
(1) CaCO3 → CaO + CO2
(2) 2KClO3 →2KCl + 3O2
(3) Digestion of food in body
(4) H2 + Cl2 →2HCl
Answer
4
Question 34. In the equation Cu + xHNO3 → Cu(NO3)2 + yNO2 + 2H2O The values of x and y are
(1) 3 and 5
(2) 8 and 6
(3) 4 and 2
(4) 7 and 1
Answer
3
Question 35. Which of the following chemical equations is an unbalanced one ?
(1) 2NaHCO3 → Na2CO3 + H2O + CO2
(2) 2C4H10 + 12O2 → + 8CO2 + 10H2O
(3) 2Al + 6H2O →2Al(OH)3 + 3H2
(4) 4NH3 + 5O2 → 4NO + 6H2O
Answer
2
Question 36. Lead nitrate on heating gives
(1) Lead oxide
(2) Nitrogen dioxide
(3) Oxygen
(4) All of these
Answer
4
Question 37. Heating limestone produces
(1) Quick lime
(2) Carbon dioxide
(3) Both (1) and (2)
(4) None of these
Answer
3
Question 38. When ferrous hydroxide reacts with hydrochloric acid, _______ and H2O are produced.
(1) FeCl3
(2) FeCl2
(3) FeCl4
(4) FeCl
Answer
4
Question 39. When copper powder is heated, it gets coated with
(1) Black copper oxide
(2) Yellow copper oxide
(3) Red copper oxide
(4) None of these
Answer
1
Question 40. Which of the following is a physical change?
(1) Solubility in water
(2) Combustibility
(3) Aerial oxidation
(4) Reaction with water
Answer
1
Whoever needs to take the CBSE Class 10 Board Exam should look at this MCQ. To the Students who will show up in CBSE Class 10 Science Board Exams, It is suggested to practice more and more questions. Aside from the sample paper you more likely had solved. These Chemical Reactions and Equations Class 10 MCQ are ready by the subject specialists themselves.
Question 41. Formation of carbon disulphide from carbon and sulphur takes place by
(1) absorption of heat
(2) evolution of heat
(3) no change in heat content
(4) None of the above
Answer
1
Question 42. In the reaction given below, a, b, c and d respectively are aFe2O3 + bH2 →cFe + dH2O
(1) 1, 1, 2, 3
(2) 1, 1, 1, 1
(3) 1, 3, 2, 3
(4) 1, 2, 2, 3
Answer
3
Question 43. Which of the following equations is representing combination of two compounds ?
(1) CaO + CO2¾→ CaCO3
(2) CO +1/2O2 ¾→ CO2
(3) SO2 +1/2O2 ¾→ SO3
(4) 2Na + 2H2O ¾→ 2NaOH + H2
Answer
1
Question 44. Which of the followin g is not a t hermal decomposition reaction ?
(1) 2H2O →2H2 + O2
(2) 2FeSO4 → Fe2O3 + SO2 + SO3
(3) ZnCO3 →ZnO + CO2
(4) 2KClO3 → 2KCl + 3O2
Answer
1
Question 45. Which of the following stands for a double displacement reaction ?
(1) 2H2 + O2 → 2H2O
(2) 2Mg + O2 →2MgO
(3) AgNO3 + NaCl → AgCl + NaNO3
(4) H2 + Cl2 → 2HCl
Answer
3
Question 46. Barium chloride on reacting with ammoniums ulphate forms barium sulphate and ammonium chloride. Which of the following correctly represents the type of the reaction involved ?
(i) Displacement reaction
(ii) Precipitation reaction
(iii) Combination reaction.
(iv) Double displacement reaction
(1) (i) only
(2) (ii) only
(3) (iv) only
(4) (ii) and (iv)
Answer
4
Question 45. What happens when dilute hydrochloric acid is added to iron filings ? Tick the correct answer.
(1) Hydrogen gas and iron chloride are produced.
(2) Chlorine gas and iron hydroxide are produced.
(3) No reaction takes place.
(4) Iron salt and water are produced.
Answer
1
Question 46. The reaction, Fe2O3(s) + 2Al(s) →Al2O3 + 2Fe(l) is an example of
(1) combination reaction
(2) double displacement
(3) decomposition reaction
(4) single displacement reaction
Answer
4
Question 47. Which of the statements about the reaction below are incorrect ?
2PbO(s) + C(s) →2Pb(s) + CO2(g)
(i) Lead is getting reduced.
(ii) Carbon dioxide is getting oxidised.
(iii) Carbon is getting oxidised.
(iv) Lead oxide is getting reduced.
(1) (i) and (ii)
(2) (i) and (iii)
(3) (i), (ii) and (iii)
(4) All of the above
Answer
1
Question 48. Which of the following is a displacement reaction ?
(1) CaCO3 → CaO + CO2
(2) CaO + 2HCl →CaCl2 + H2O
(3) Fe + CuSO4 → FeSO4 + Cu
(4) NaOH + HCl →NaCl + H2O
Answer
3
Question 49. The chemical formula for slaked lime is
(a) Ca(OH)2
(b) CaCO3
(c) CaO
(d) CaHCO3
Answer
A
Question 50. The chemical formula of lead nitrate is:
(a) KNO3
(b) PbNO3
(c) K(NO3)2
(d) Pb(NO3)2
Answer
D
Question 51.When green coloured ferrous sulphate crystals are heated, the colour of the crystal changes because:
(a) it is decomposed to ferric oxide
(b) it loses water of crystallisation
(c) it forms SO2
(d) it forms SO3
Answer
B
Question 52 .What is observed when a solution of potassium iodide is added to silver nitrate solution?
(a) No reaction takes place
(b) White precipitate of silver iodide is formed
(c) yellow precipitate of Agl is formed
(d) Agl is soluble in water.
Answer
C
Question 53.The reaction between lead nitrate and potassium iodide present in aqueous solutions is an example of visit for Topper’s materials
(a) Decomposition Reaction
(b) Displacement Reaction
(c) Double Displacement Reaction
(d) Neutralisation Reaction
Answer
C
Question 54. An iron nail was dipped in a salt solution. After sometime a reddish brown deposition of the nail was seen. The salt solution could be
(a) Silver nitrate
(b) Sodium sulphate
(c) Aluminium chloride
(d) Copper sulphate
Answer
D
Question 55.PbS reacts with ozone (O3) and forms pbso4 . As per the balanced equation, molecules of ozone required for every one molecule of PbS is/are
(a) 4
(b) 3
(c) 2
(d) 1
Answer
A
Question 56 .The condition produced by aerial oxidation of fats and oils in foods marked by unpleasant smell and taste is called:
(a) Antioxidation
(b) Reduction
(c) Rancidity
(d) Corrosion
Answer
C
Question 57. Which of the following reactions will not take place?
(a) Zn + CuSO4→ ZnSO3+ Cu
(b) 2KBr + Cl2→KCI+ Br2
(c) Zn + MgSO4→ ZnSO4+ Mg
(d) Mg + FeSO4– MgSO4+ Fe
Answer
(c) Zn + MgSO4→ ZnSO4+ Mg
Answer
C
Question 58. Which of the following is a thermal decomposition reaction?
(a) 2H2O → 2H2+ O2
(b) 2AgCl → 2Ag + Cl2
(c) H2(g) + Cl2(g) → 2HCl(g)
(d) ZnCO3→ ZnO + CO2
Answer
D
Question 59. The colour of the residue left in the test tube after heating ferrous sulphate which undergoes decomposition is
(a) yellowish-brown
(b) black
(c) white
(d) grey
Answer
A
Question 60 .Chemically rust is
(a) Hydrated ferrous oxide
(b) hydrated ferric oxide
(c) only ferric oxide
(d) none of these
Answer
B
Question 61. The chemical formula for lime is
(a) Ca(OH)2
(b) CaCO3
(c) CaO
(d) CaHCO3
Answer
C
Question 62. The reaction of water and quick lime is an example of
(a) combination reaction
(b) exothermic reaction
(c) both (a) and (b)
(d) None of these.
Answer
C
Question 63. Reaction between calcium oxide and water is a ______________ reaction.
(a) endothermic reaction
(b) decomposition reaction
(c) exothermic reaction
(d) displacement reaction
Answer
C
Question 64 . The compound also known as blue vitriol is
(a) FeSO4.7H2O
(b) CuSO4.5H2O
(c) CaSO4.2H2O
(d) Na2CO3.10H2O
Answer
B
Question 65. Which of the following is an endothermic reaction?
(a) CaCO3 heat CaO + CO2
(b) CaO + H2O Ca(OH)2
(c) C6H12O6 + O2 6CO2 + 6H2O
(d) None of these
Answer
A
Question 66: In the double displacement reaction between aqueous potassium iodide and aqueous lead nitrate, a yellow precipitate of lead iodide is formed. While performing the activity if lead nitrate is not available, which of the following can be used in place of lead nitrate?
(a) Lead sulphate (insoluble)
(&) Lead acetate
(c) Ammonium nitrate
(d) Potassium sulphate
Answer
B
Question 67. How many water molecules are present in a crystal of copper sulphate molecule?
(a) 5
(b) 7
(c) 2
(d) 3
Answer
A
Question 68. How many water molecules are present in a crystal of ferrous sulphate molecule?
(a) 5
(b) 2
(c) 7
(d) 10
Answer
C
Question 69: Which among the following statement(s) is (are) true? Exposure of silver chloride to sunlight for a long duration turns grey due to
(i) the formation of silver by decomposition of silver chloride
(ii) sublimation of silver chloride
(iii decomposition of chlorine gas from silver chloride
(iv) oxidation of silver chloride
(a) (i) only
(b) (i) and (iii)
(c) (ii) and (iii)
(d) (iv) only
Answer
A
Question 70: The condition produced by aerial oxidation of fats and oils in foods marked by unpleasant smell and taste is called:
(a) antioxidation
(b) reduction
(c) rancidity
(d) corrosion
Answer
C
Question 71: Select the oxidising agent for the following reaction:
H2S + I2 > 2HI + S
(a) I2
(b) H2S
(C) HI
(d) S
Answer
A
Question 72: A substance added to food containing fats and oils is called:
(a) Oxidant
(b) Rancid
(c) Coolant
(d) Antioxidant
Answer
D
Question 73: When SO2 gas is passed through saturated solution of H2S, which of the following reaction occurs?
(a) SO2 + 2H2S → 2H2O + 3S
(b) SO2 + 2H2S → H2O + 3S
(c) SO2 + H2S → H2O + S
(d) SO2 + H2O → SO3 + H2
Answer
A
Question 74: Pb + CuCl2 → PbCl2 + Cu
The above reaction is an example of:
(a) combination
(b) double displacement
(c) decomposition
(d) displacement
Answer
D
Question 75: Which of the following gases can be used for storage
(a) Carbon dioxide or Oxygen
(b) Nitrogen or Oxygen
(c) Carbon dioxide or Helium
(d) Helium or Nitrogen
Answer
D
Question 76: Electrolysis of water is a decomposition reaction. The mole ratio of hydrogen and oxygen gases liberated during electrolysis of water is:
(a) 1 : 1
(b) 2:1
(c) 4:1
(d) 1:2
Answer
B
Question 77:Name the products formed when iron filings are heated with dilute hydrochloric acid
(a) Fe (III) chloride and water
(b) Fe (II) chloride and water
(c) Fe (II) chloride and hydrogen gas
(d) Fe (III) chloride and hydrogen gas
Answer
D
Question 78. What is the colour of FeSO4.7H2O?
(a) Blue
(b) Green
(c) White
(d) Brown
Answer
B
Question 79: What type of chemical reactions take place when electricity is passed through water?
(a) Displacement
(b) Combination
(c) Decomposition
(d) Double displacement
Answer/ Explanation
Answer
C
You can easily get good marks If you study with the help of Class 10 Chemical Reactions and Equations MCQ. We trust that information provided is useful for you. NCERT MCQ Questions for Class 10 Chemical Reactions and Equations PDF Free Download would without a doubt create positive results.
We hope the information shared above in regards to MCQ on Chemical Reactions and Equations Class 10 with Answers has been helpful to you. if you have any questions regarding CBSE Class 10 Science Solutions MCQs Pdf, write a comment below and we will get back to you as soon as possible.
Frequently Asked Question (FAQs)
How many MCQ questions are there in Class 10 Chapter 1 Science?
In Class 10 Chapter 1 Science, we have provided 79 Important MCQ Questions, But in the future, we will add more MCQs so that you can get good marks in the Class 10 exam.
Can we score good marks in Class 10 Science with the help of Chemical Reactions and Equations MCQ Questions?
Yes, MCQ Question is one of the best strategies to make your preparation better for the CBSE Board Exam. It also helps to know the student’s basic understanding of each chapter. So, You can score good marks in the Class 10 Science exam.


